What Is A Fuel Cell
In layman's terms, a Fuel Cell is a Hydrogen Battery. But what is a hydrogen battery? Many people are familiar with the lead-acid battery used in cars. Your car battery uses acid and lead to produce electricity. A hydrogen battery, or fuel cell if you want to get fanciful, uses hydrogen as its main ingredient to produce electricity.
 |
The definition of a fuel cell is simply a device which uses an electrochemical process to produce electrical power. In its simplest form, your car battery is a fuel cell. But there has been a lot of talk lately about fuel cell technology and how exciting this can be for the automotive industry. This excitement hardly refers to the existing lead acid battery used in cars today.
Although scientifically a battery is considered a fuel cell, but the fuel cell terminology used in the automotive industry actually refers to a far more efficient device which produces electricity through the chemical reaction between hydrogen and oxygen. Many types of fuel cell exists, and they are classified according to the type of electrolyte they use.
Essentially, six components are needed in a fuel cell – a cathode, an anode, an electrolyte, hydrogen, oxygen and a catalyst.
High pressure hydrogen and oxygen is injected into the fuel cell where it reacts with the catalyst which is usually platinum powder. The reaction produces positive and negative electrons which are conducted out to an external circuit through the anode and cathode. The hydrogen atoms which are positively charged are then attracted to the negatively charged oxygen atoms. They combine together and produce water. The electrolyte in the fuel cell functions to facilitate the transfer of electrons.
The final result of the electrochemical reaction is water, a little heat and a small amount of electrical energy. Higher electrical output can be achieved by the simultaneous reaction of hydrogen and oxygen all occurring at the same time. In a fuel cell vehicle such as the Honda FCX, an external storage tank is used to store liquid hydrogen, whereas oxygen is obtained from atmospheric air. Because the reaction is highly efficient, a single tank can provide vast amounts of energy before needing to be replenished. This makes fuel cells ideal for powering electric cars, where they are not only durable, but also environmentally friendly.
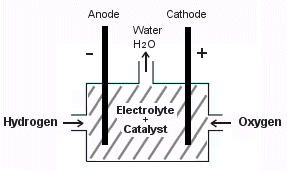 |
| A Hydrogen Fuel Cell Diagram |
Challenges With Fuel Cells
The problem with fuel cells is liquid hydrogen is not so readily available. You can’t drive to your local gas station and fill up on liquid hydrogen. But even if the infrastructure exists, hydrogen itself is not so easy to extract. Although hydrogen exists naturally in water, but current methods of extraction requires more energy to extract hydrogen from water than the energy it produces. While it is much easier to extract hydrogen from hydrocarbon fuels (such as gasoline), but this method produces pollution as a by product. Since the primary goal of fuel cell is to reduce pollution, then this method is not acceptable.
The second challenge facing fuel cells is in the catalyst. Precious metals such as platinum is used and there is not enough platinum in the entire world to fill up a room the size of your bedroom.
Fuel cell technology is still in the early development phase and until such time when these challenges are overcome, fuel cells will remain prohibitively expensive. The current cost of a fuel cell runs into several million dollars to produce. As you might have learnt, fuel cells have nothing to do with Hydroxene or gasoline-hydrogen hybrid cars.
|
We apologize but this website is still being updated with more content about Hydroxene and Hybrid Cars. Some or most of the links here do not work yet, but they are being completed as quickly as time permits.
If you encounter a broken link, then please re-visit us again about a week later. The last update is on December 29, 2006. When the website is complete, this message will be removed. |
